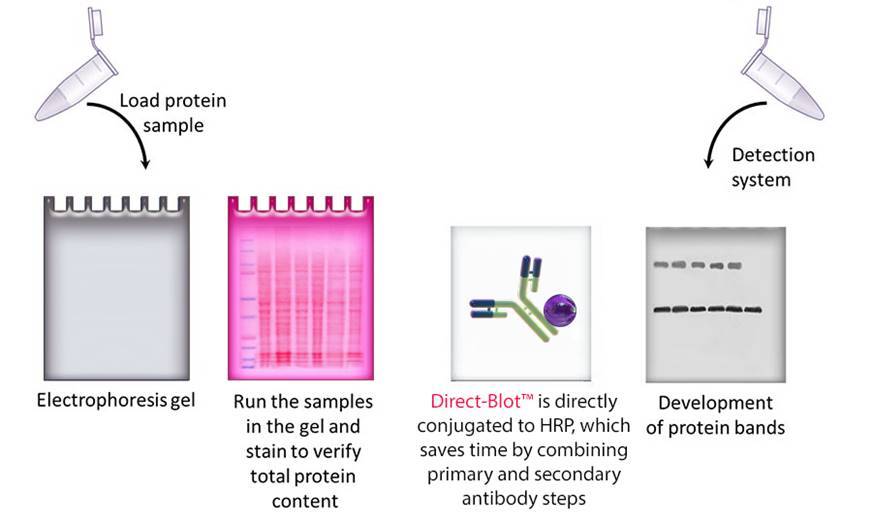
The method details how to extract the protein from the tissue, followed by separation of the proteins by size using sodium dodecyl sulfate (SDS) polyacrylamide-gel electrophoresis (PAGE) and transfer of the proteins to a membrane to allow immunodetection of the iNOS. This chapter describes a method for the estimation of the amount of iNOS in tissue samples however, it is easily adapted for the other forms of NOS by the use of the appropriate antibodies. These antibodies are specific for the particular isoforms of NOS and show no cross-reactivity with other isoforms, identifying proteins of apparent Mr 130 kDa, 160 kDa, and 133 kDa respectively (1). Antibodies are now commercially available for the detection of the inducible (i), neuronal (n), and endothelial (e) NOS. The amount of nitric oxide synthase (NOS) in tissues and cells can readily be determined by Western blotting. After washing off excess unbound secondary antibody, the amount of the secondary antibody bound to the membrane is proportional to the amount of the original antigen present in the cell extract and can be determined by the activity of the enzyme to which it is linked, e.g., by enhanced chemiluminescence (ECL). After a series of washes to remove the excess unbound antibody, the membrane is then exposed to a secondary antibody conjugated to an enzyme, which specifically recognizes the primary antibody. In order to detect the enzyme on the immunoblot, the blotted-cellular proteins are incubated with a primary antibody specific for the protein to be estimated, e.g., iNOS-polyclonal antiserum, to allow the antibody to bind specifically to its antigen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
